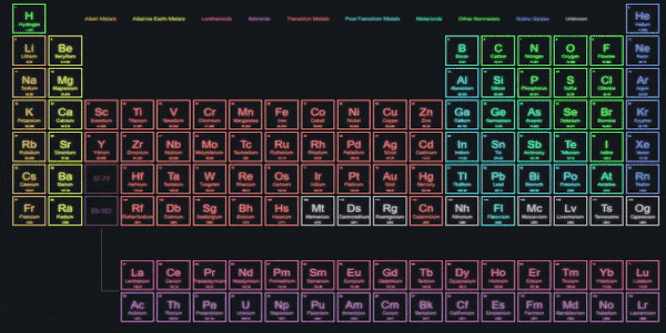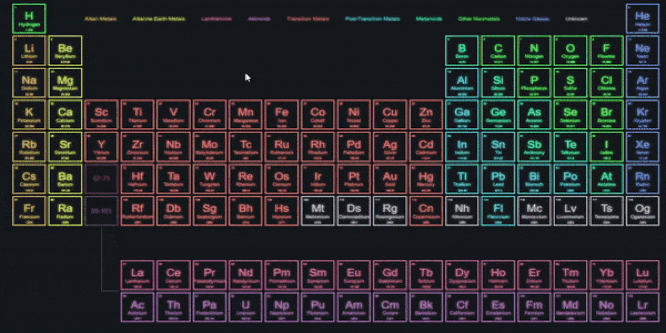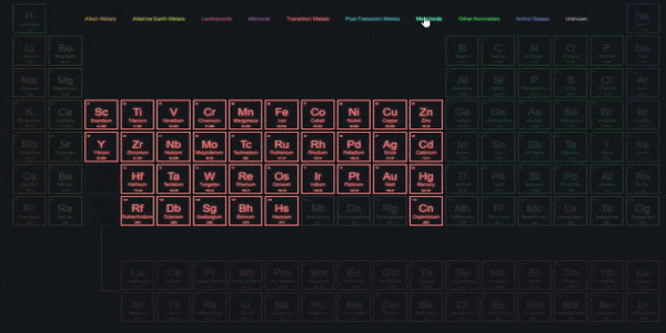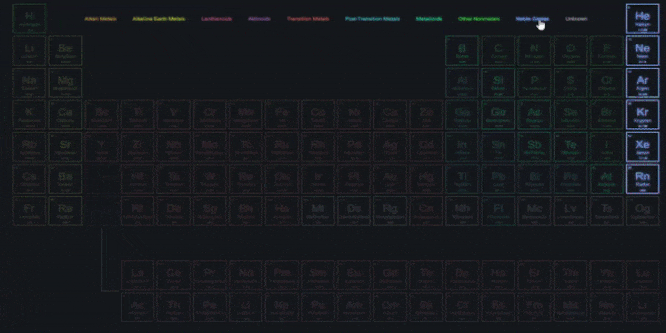
With similar properties were grouped together, 1. A systematic study of elements Elements.Elements in the same group exhibit similarĪchievements of Mendeleevs Periodic Table.Groups VIII doesnt have any subgroups and. 
Groups I to VII subdivided into A and B.Eight vertical columns known as groups.Twelve horizontal rows, which were condensed to.The physical and chemical properties of elementsĪre periodic functions of their atomic weights.He also grouped unlike elements under the same.The same slot to fit elements in the table. In a few cases, Newlands placed two elements in.Properties of new elements discovered did not fit.The law was applicable for elements with atomic.Placed some unlike elements in the same slot. Which is a drawback of Newland Law of Octaves. According to him, only 56 elements are found.That every eight element has same properties as It says that elements are arranged in such a way.Three triads from the elements known at that time. But, Dobereiner could identify only following.He showed that the atomic mass of the middleĮlement is approximately the arithmetic mean of.Increasing atomic masses and called it a triad. Dobereiner arranged a group of three elements.Elements with similar properties are placed in.Properties of different elements in a periodic Classification is needed to easily understand the.Placing similar groups and species together is.All of these have different properties.There are about 118 elements out of which 94.Matter around us are present in the form of.Sincerely, Ryan Murphy M.Title: Periodic classification of elements If you have any questions please feel free to contact me. This unit aligns with the Next Generation Science Standards and with Common Core Standards for ELA and Literacy for Science and Technical Subjects. Also included is a 190 slide first day of school PowerPoint presentation.Īreas of Focus: -Atoms (Atomic Force Microscopes), Rutherford's Gold Foil Experiment, Cathode Tube, Atoms, Fundamental Particles, The Nucleus, Isotopes, AMU, Size of Atoms and Particles, Quarks, Recipe of the Universe, Atomic Theory, Atomic Symbols, #'s, Valence Electrons, Octet Rule, SPONCH Atoms, Molecules, Hydrocarbons (Structure), Alcohols (Structure), Proteins (Structure), Periodic Table of the Elements, Organization of Periodic Table, Transition Metals, Electron Negativity, Non-Metals, Metals, Metalloids, Atomic Bonds, Ionic Bonds, Covalent Bonds, Metallic Bonds, Ionization, and much more. This unit also includes four PowerPoint review games (110+ slides each with Answers), 38+ video links, lab handouts, activity sheets, rubrics, materials list, templates, guides, and much more. Also included are critical class notes (color coded red), project ideas, video links, and review games. The PowerPoint includes built-in instructions, visuals, and review questions. The activities and discussion questions in the slideshow are meaningful. The answer keys and unit notes are great for support professionals. The homework and lesson notes chronologically follow the PowerPoint slideshow. This PowerPoint is one small part of the Atoms and Periodic Table of the Elements unit from This unit consists of a five part 2000+ slide PowerPoint roadmap, 12 page bundled homework package, modified homework, detailed answer keys, 15 pages of unit notes for students who may require assistance, follow along worksheets, and many review games.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
